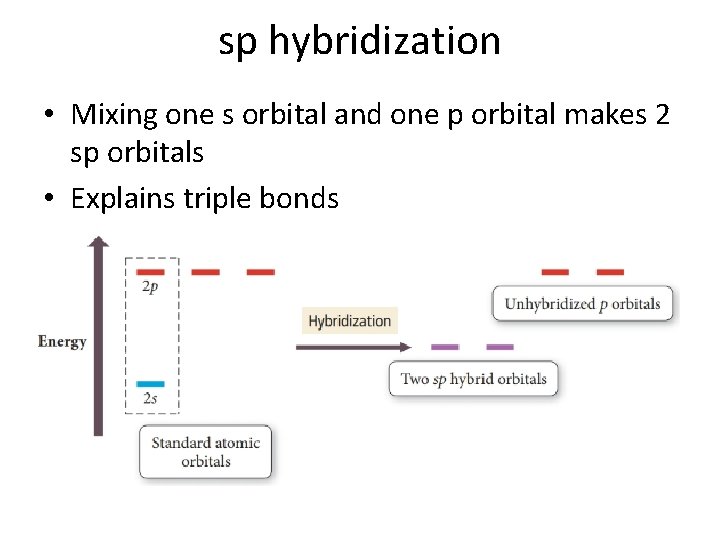
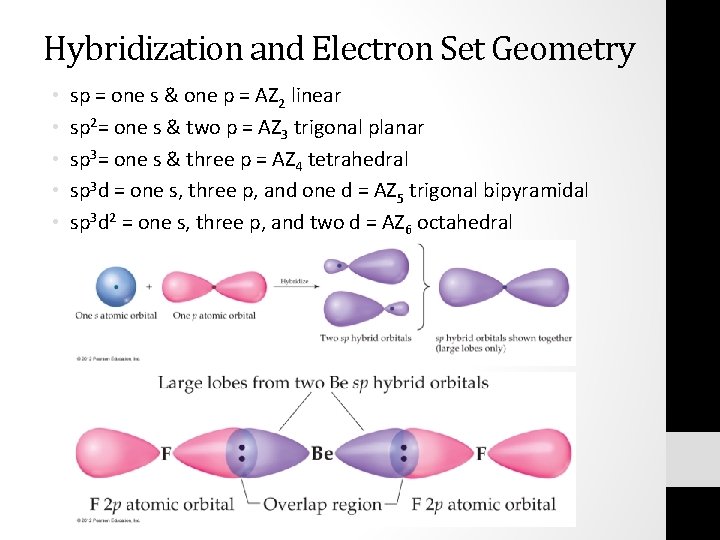
Sp Hybridization In the case of the sp hybridization, only one s and one porbital are mixed together to make hybrids This leaves two unused porbitals The unused sp orbitals force the structure to have a linear 3D geometry The unused porbitals can make two double bonds, a triple bond, or potentially participate in resonance with otherSp mixing causes the 2p So H–F has one σ bond and three lone electron pairs on fluorine Relative AO Energies for MO Diagrams H He Li Be B C N O F Ne B C N O F Ne Na Mg Al Si P S Cl Ar Al Si P S Cl Ar 1s 2s 2p 3s 3p –194 eV –158 eV –324 eV –107 eV Photoelectron spectroscopy gives us a pretty good idea of the relativeLog into Facebook to start sharing and connecting with your friends, family, and people you know

Pzl Fighters Part One
Sp one products
Sp one products-Sp 2 hybridization is observed when one s and two p orbitals of the same shell of an atom mix to form 3 equivalent orbitals The new orbitals formed are called sp 2 hybrid orbitals sp 2 hybridization is also called trigonal hybridizationSP Flash Tool Download Smart Phone Flash Tool SP Flash Tool (SmartPhone Flash Tool) helps you flash or install the Stock Firmware (scatter based) on your Mediatek powered Smartphone and Tablet It also allows you to flash a custom recovery, Format the Device, Reset the Device and Remove FRP Protection




What Number And Type Of Hybrid Orbital S Form S When One P Clutch Prep
Modify your Jeep, truck, muscle car or offroad vehicle with cylinders, tierods and other high quality steering components from PSC MotorsportsEquivalent sp2 orbitals, leaving one p orbital untouched The process is shown below 2s 2p X 2p y 2p z Potential energy sp2 hybridization sp2 sp2 sp2 p In this top view, the unhybridized p orbital cannot be seen because it also arranges itself to be as far apart from the sp2 orbitals as possible That is to say, it is positioned at rightThere are two remaining p orbitals Next we show the phase pictures of combining the sp hybrid orbitals with fi rst one and then both of the remaining p orbitals Phase pictures loop Diagram Axes for you to draw the loop diagram You will also see these orbitals
P ORBITALS Not all electrons inhabit s orbitals At the first energy level, the only orbital available to electrons is the 1s orbital However, at the second level, there are also orbitals called 2p orbitals in addition to the 2s orbital Unlike an s orbital, a p orbital points in a particular direction The one shown below points up and downThe three p orbitals differ in the way the lobes are oriented whereas they are identical in terms of size shape and energy As the lobes lie along one of the x, y or zaxis, these three orbitals are given the designations 2p x, 2p y, and 2p z Thus, we can say that there are three p orbitals whose axes are mutually perpendicularPortuguese for 'Saint Paul') is a city in the Southeast Region of BrazilListed by the GaWC as an alpha global city, the municipality of São Paulo is the most populous city proper in Brazil, the Americas, the Western Hemisphere and the Southern Hemisphere, as well as the world's 4th largest city proper by population
Bringing an app store experience to IU servicesShare photos, videos &The S&P 500 index is a basket of 500 large US stocks, weighted by market cap, and is the most widely followed index representing the US stock market S&P 500 Monthly Return is at 476%, compared to 290% last month and 392% last year This is lower than the long term average of 053%
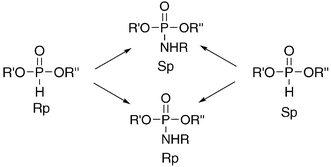



Controlling Stereochemistry During Oxidative Coupling Preparation Of Rp Or Sp Phosphoramidates From One P Chiral Precursor Chemical Communications Rsc Publishing




Astell Kern Acro L1000 Celsus Sound Sp One P Kupit V Kieve I Ukraine Po Cene Grn Na Portativ Ua
Best LowLatency Data Feed Provider;A) one s, one p b) two s, one p c) two s, two p d) one s, two p e) zero s, two p Answer a 6 In order to form a set of sp 3 d hybrid orbitals, how many pure atomic orbitals must be mixed?Unhybridized p orbitals allow for pi bonding, not empty hybridized orbitals Take for example an sp2, you would have the 3 hybridized sp2 orbitals, and then remember you would still have a leftover p orbital (you had 4 total 1 s and 3 p's), so that leftover p orbital can make the pi bond, resulting in a double bond




Valence Bond Theory Hybridization Pdf Free Download




Sp Tyre Service Mahkota Cheras昇萍輪胎服務 Home Facebook
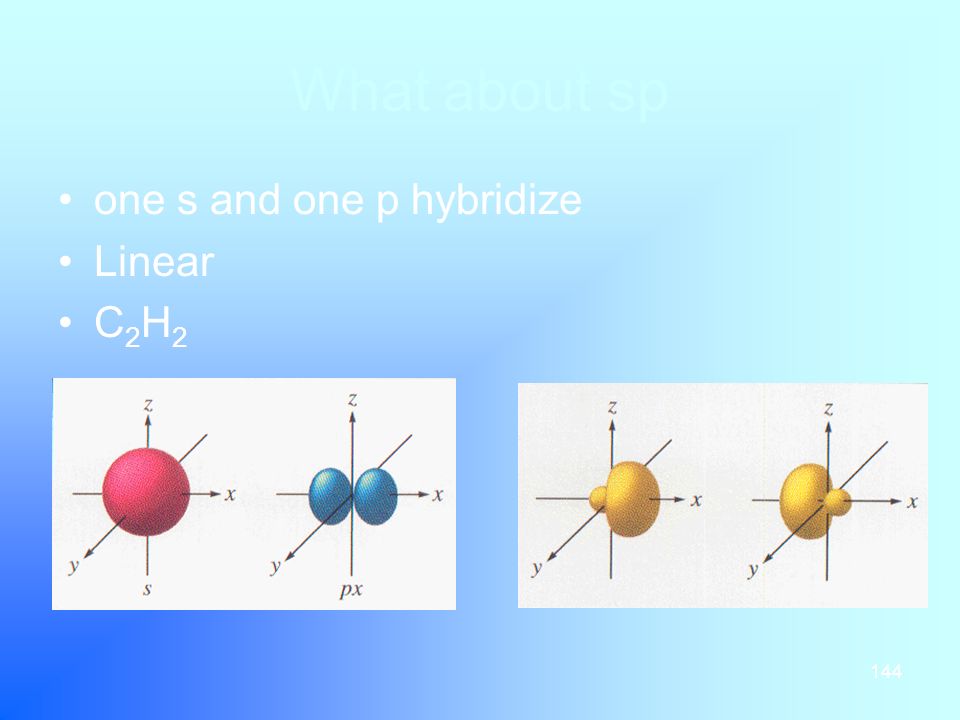
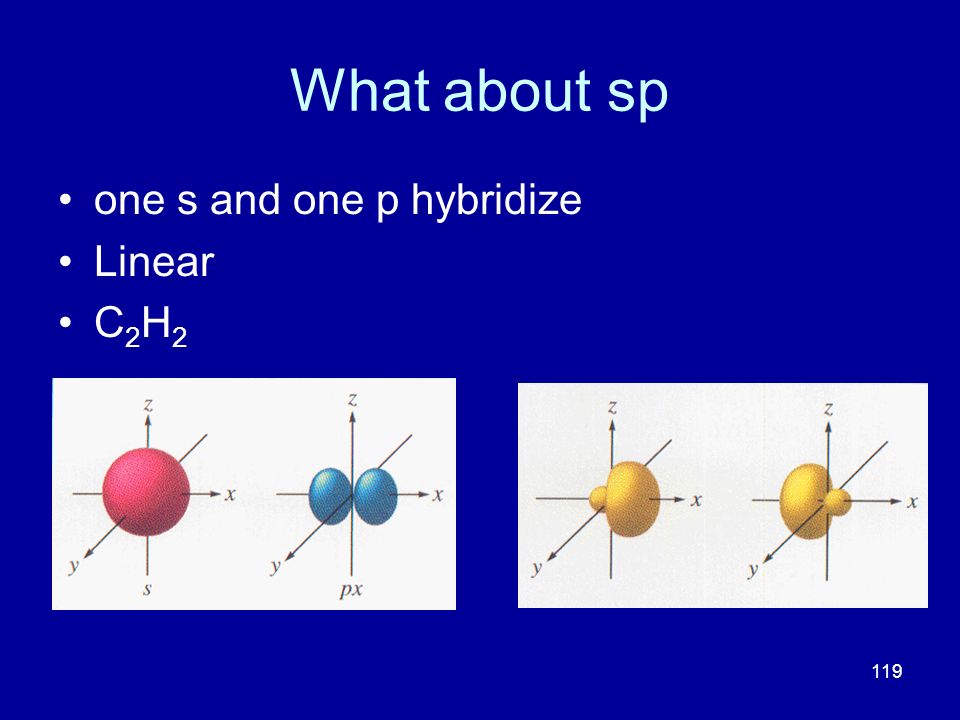
Investors' focus this week will be on earnings results, with some of the most heavily weighted companies in the S&P 500 poised to deliver their quarterly reports There's a housing boom going onOne of the best things about S&P 500 ETFs is that they're passive investments, and you can supercharge your savings by simply leaving your money alone Historically, the S&P 500 itself has earnedAn sp hybridized atom uses one s and one p orbital to make two sp hybrid orbitals;



Gray Anchor Roma 10a S P One Way Switch Module Size 1 Module 230 V Rs 30 Piece Id



Plos One Expression Of A Chimeric Antigen Receptor In Multiple Leukocyte Lineages In Transgenic Mice
Create an account or log in to Instagram A simple, fun &Transcript In sp hybridization, one s orbital and one p orbital hybridize to form two sp orbitals, each consisting of 50% s character and 50% p character This type of hybridization is required whenever an atom is surrounded by two groups of electronsMessages with friends &




画像をダウンロード Sp Onep 無料のワンピース画像



フューレンコーディネート Celsus Soundのスピーカー Celsus Sp One の発売を発表 Mj 無線と実験
Hybridization, one s orbital and two p orbitals hybridize to form three sp²Get a complete List of all S&P 500 stocks The values of #name# companies consists live prices and previous close price, as well as daily, 3, 6Current and Historical Performance Performance for SPDR S&P 500 on Yahoo Finance




Abarth Una Splendida One Off Omaggia La 1000 Sp Degli Anni Sessanta La Stampa




Dreer S Garden Book 19 19 Dreer S Garden Book 19 Dreersgardenbook19henr Year 19 16 F Lom Cucvj A A A A V3ii Ua Lt swal Ji I I A G3ic Ti A R Ah8 Fe5 A A T Fa Saaww Igsa2as A G A K I M P
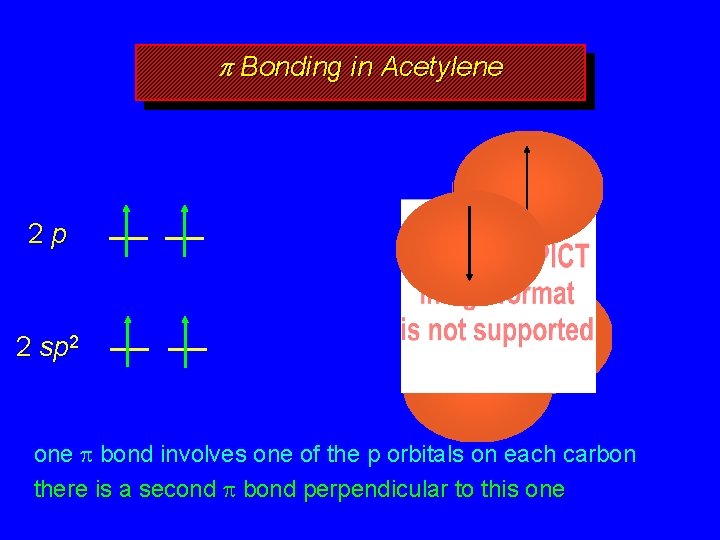
In an #sp^3# hybridization, #color(red)one# #s# orbital is mixed with #color(red)three# #p# orbitals to form #color(red)four# #sp^3# hybridized orbitals Each of these hybridized orbitals have 25% s character and 75% p character (calculated according to the proportion of sp mixing) These #sp^3# hybridized orbitals are oriented with bond angle ofGo to the Windows 7 Service Pack 1 download page on the Microsoft website Select Install Instructions to see which packages are available for download, and make note of the one that you need Select the appropriate language from the dropdown list, and then select Download Select the packages you need to install, select Next, and then followThus, the ethylene molecule is said to have sp2s bonds (4 CH bonds) , one sp 2sp 2 bond (CC bond) and one pp pi bond (CC bond) Thus, the sp 2 hybridization theory explains the double bond, the trigonal planar structure in ethylene molecule Similarly, for a triple bond formation, like for that of acetylene molecule, there is sp



Civicimuseiudine It




One Hybridization Of One S And One P Orbital We Get
Access Google Drive with a free Google account (for personal use) or Google Workspace account (for business use)Index performance for S&P 500 INDEX (SPX) including value, chart, profile &São Paulo (/ ˌ s aʊ ˈ p aʊ l oʊ /, Portuguese sɐ̃w̃ ˈpawlu ();




Automatic One Layer Cutter Cutting Width 170 Cm Texi Xyz Sp 170




Zerodol Sp Tablet 10 S Buy Medicine Online At Best Price Netmeds
Sp 2 Hybridization The valence orbitals of a central atom surrounded by three regions of electron density consist of a set of three sp 2 hybrid orbitals and one unhybridized p orbital This arrangement results from sp 2 hybridization, the mixing of one s orbital and two p orbitals to produce three identical hybrid orbitals oriented in a trigonal planar geometry ()Search the world's information, including webpages, images, videos and more Google has many special features to help you find exactly what you're looking forProcedures (SP&P) Section (1) (b)1, Florida Statutes (FS), requires that district school boards submit to the Florida Department of Education (FDOE) proposed procedures for the provision of special instruction and services for exceptional students once every three years Approval of this document by FDOE is required by



Chapter 10 Chemical Bonding Molecular Shapes Valence Bond




Mixed Strategy Nash Equilibrium For The One Shot S P Game Download Scientific Diagram
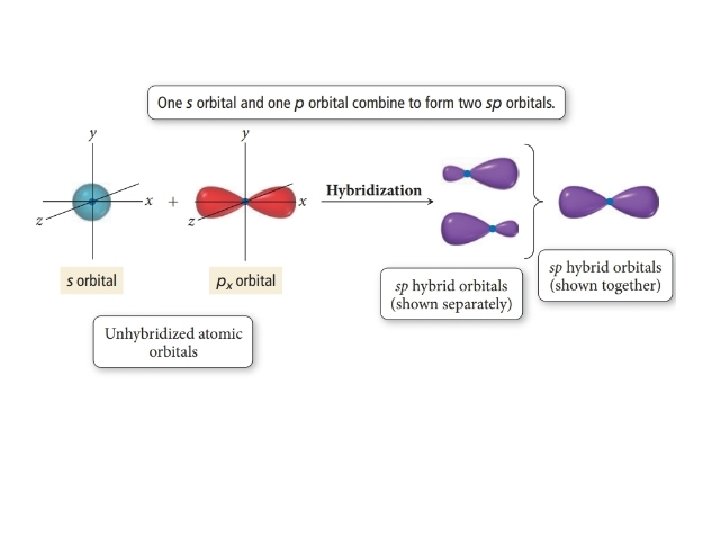
SPS Corporate Website Per IRS regulations, all 1098 yearend statements will be mailed no o later than January 31st Yearend information will be available via our automated phone system on January 2nd, 21 Yearend statements can be obtained from our website after February 10th, 21 Log into your account to view or print a copy ofIn sp hybridization, the s orbital overlaps with only one p orbital Atoms that exhibit sp hybridization have sp orbitals that are linearly oriented;An sp^3 orbital is unsymmetrical in shape, having one small and one large lobe The four sp^3 hybrid orbitals of a group are equivalent in shape and energy A central atom can be surrounded by five or six electron groups if _____ orbitals are available for bonding




Annotationes Zoologicae Japonenses Nihon Do Butsugaku Iho New Or Imperfectly Known Species Of Earthworms 15 P Shimaensis N Sp One Specimen Preserved In Formol Length 5 Mm Breadth 7 Mm




Skirted One Piece Dual Flush 3 4 5l Washdown Toilet With P Trap 453x Sp Kohler
S&P Global Sign In New User Sign Up Use your existing S&P Global login credentials to access Marketplace Otherwise, please enter your email address below to begin the registration process You may gain access to this product if your current company has an active Master Subscription Agreement with S&P Global Market IntelligenceOpen SAP ONE Support Launchpad Visit SAP Support Portal or SAPcomQuestion The blending of one s atomic orbital and one p




Sedile Vw 085 4th Cambio 41 45 Denti d b 695 Eur 124 98 Picclick It



Plos One Seed Biopriming With P And K Solubilizing Enterobacter Hormaechei Sp Improves The Early Vegetative Growth And The P And K Uptake Of Okra Abelmoschus Esculentus Seedling
Up to32%cash backShoe Palace prides itself on offering its customers the Ultimate Experience when it comes to their shoe and apparel shopping Its locations, staff, and products are all the very best imaginable Shoe Palace carries only elite shoe and apparel brands Its chic and loyal clientele reflect that commitment to retail excellence Simple is superiorTwo sp orbitals will be at 180 degrees to each other Any central atom surrounded by just two regions of valence electron density in a molecule will exhibit sp hybridization Some examples include the mercury atom in the linear HgCl 2 moleculeCreative way to capture, edit &




Volkswagen Da Gear 0 02k 5th Di Trasmissione Ingranaggio 38 51 Denti Da Gear Ebay




Dreer S Garden Book 1921 1921 Dreer S Garden Book 1921 Dreersgardenbook1921henr Year 1921 26 L1lehrradreera Philadelpt1laa A Wreliabletoetable Seeds Tu Aubergine Fr P Aj Tt Berengena Sp Eierpflanze Ger Chicoree Fr Endive Endime
Each sp hybrid orbital has one large and one small lobe Hybrid orbitals are designated by using a superscript to indicate the _____ of each type of orbital used to form them For example, the designation sp^2 indicates that _____ s and _____ p orbital(s) were combined to form this orbital typeRecent awards include Best Credit Risk Management Product;A) one s, one p, and one d b) one s, three p, and one d c) two s, one p, and two d d) two s, six p, and two d



More Marineford Characters Join One Piece Unlimited Cruise Sp Siliconera




Sp One P Celsus Sound Hifi Do Mcintosh Jbl Audio Technica Jeff Rowland Accuphase
The unhybridized atomic p orbital lies at a 90°17年2月日月曜日 おすすめの組合せ。 省スペースでも高音質、YAMAHAの『WXA50』&Celsus Sound『SPONE/P』。 『Music Cast』体験イベントにご用意させて頂いた、YAMAHAの『WXA50』。 コンパクトな筐体に、DSD56MHz再生にも対応したネットワークプレーヤーと、 定格Sign in with your organizational account Look up your email address / student ID Reset Password Create Password



Plos One P Coumaric Acid Influenced Cucumber Rhizosphere Soil Microbial Communities And The Growth Of Fusarium Oxysporum F Sp Cucumerinum Owen




Pzl Fighters Part One
SAP ONE Support Launchpad What would you like to do next?Orbitals, each consisting of 33% s character and 67% p character This type of hybridization is required whenever an atom is surrounded by three groups of electronsIf your company has a current subscription with S&P Global Market Intelligence, you can register as a new user for access to the platform(s) covered by your license at S&P Capital IQ Pro or S&P Capital IQ



Plos One P Coumaric Acid Influenced Cucumber Rhizosphere Soil Microbial Communities And The Growth Of Fusarium Oxysporum F Sp Cucumerinum Owen




Poland S Pzl Gull Wing Fighters Volume One P 1 Through P 8 Warren A Eberspacher Jan P Koniarek Amazon Com Books
Angle to the plane This configuration allows for the maximum separation of all orbitals Last, the atomic orbitals of carbon can hybridize by the linear combination of one s and one p orbital This process forms two equivalent sp hybrid orbitals The remaining two atomic p orbitals remainDow Jones The Dow Jones branded indices are proprietary to and are calculated, distributed and marketed by DJI Opco, a subsidiary of S&P Dow Jones Indices LLC and have been licensed forThe blending of one s atomic orbital and one p atomic orbital produces _____ a two sp hybrid orbitals b two sp2 hybrid orbitals c three sp2 hybrid orbitals d two sp3 hybrid orbitals e three sp hybrid orbitals;




Celsus Sound Sp One Active And Passive Audio Graffiti




Celsus Sound Sp One Active And Passive Audio Graffiti
For the hydrogen fluoride molecule, for example, two F lone pairs are essentially unhybridized p orbitals, while the other is an sp x hybrid orbital An analogous consideration applies to water (one O lone pair is in a pure p orbital, another is in an sp x hybrid orbital) See also Crystal field theory;Sp2 hybridization in ethene In sp^2 hybridization, the 2s orbital mixes with only two of the three available 2p orbitals, forming a total of three sp^2 orbitals with one porbital remaining The two carbon atoms form a sigma bond in the molecule by overlapping two sp 2 orbitals Each carbon atom forms two covalent bonds with hydrogen by s–spSP Flash tool download is the best application to flash Stock firmware, Custom recovery and fix some extreme issues on MediaTek Android smartphones Download SP Flash tool all versions from our official download page




5 The Sp 2 Orbitals Are Formed From One S And Two P Orbitals One P Download Scientific Diagram




Sp One P Celsus Sound Hifi Do Mcintosh Jbl Audio Technica Jeff Rowland Accuphase




Bookshelf Speakers Review Shootout Celsus Sound Sp One Vs Centrance Masterclass 2504 Vs John Blue Jb3 Headphone Reviews And Discussion Head Fi Org



18 2 Bonding In Methane And Orbital Hybridization




Solved Question 21 1 Pts What Are The Orbitals On The Chegg Com




L Abarth 1000 Sp Degli Anni Sessanta Ispira Una One Off Evocativa




Sch4u1 September Mr Dvorsky Ppt Download




Winchester 30 06 180 Gr Power Point Sp One Full One Partial 35 Rounds




Pygidial Plate Of One Of The Paratypi Of P Romanoi N Sp With Weak Download Scientific Diagram



Factory Price Hiv Rapid Test Kits Anti Hiv 1 2 Test Buy Hiv Rapid Test Hiv Blood Test Kit Hiv Test Kit Product On Alibaba Com




Sp One P Celsus Sound Hifi Do Mcintosh Jbl Audio Technica Jeff Rowland Accuphase



Plos One P Coumaric Acid Influenced Cucumber Rhizosphere Soil Microbial Communities And The Growth Of Fusarium Oxysporum F Sp Cucumerinum Owen




Review Celsus Sound Sp One P 좋은 소리를 만들겠다는 의지가 가득 담긴 작은 스피커 네이버 블로그




1 X Snr O E Opel Vauxhall Pf6 Gearbox Bearing Acquisti Online Su Ebay




A Sp Hybridisation One S Orbital Mixes With One P Orbital Forming Two Equivalent Sp Hybrid Orbitals Types Of Hybridisation Linear 180 Two Sp Hybrid




Drum Machine Moduli Trigger Batterie




Porcelanosa Sp One Xl Bathtub Tiles And Bathrooms Online




A Hysteresis Cycle For A Thought Intrusion Extrusion Experiment On Download Scientific Diagram




What Number And Type Of Hybrid Orbital S Form S When One P Clutch Prep



Plos One The Novel Reversible Lsd1 Inhibitor Sp 2577 Promotes Anti Tumor Immunity In Switch Sucrose Nonfermentable Swi Snf Complex Mutated Ovarian Cancer




Monkey Salt And Pepper Balancing Act One Pc Set Monkey S P




Audio Square Fujisawa 話題の小型スピーカー Celsus Sound セルサス サウンド Sp One P を期間限定でお聴き頂けます



Baby Silicone Plate Bibs Bowl Spone Feeding Tableware Waterproof Non Slip Crockery Bpa Free Silicone Dishes For Baby Bowl Baby P Dishes Aliexpress




Genuine Gratis Lander Ird Guarnizione Kit Anello Sp Land Rover Ebay




Arena Sp Der One P Ece Girls Swimwear Superdokan




Giunto Aria Condizionata Compressore Per Skoda Vw Audi Sedile 1jm Eur 86 42 Picclick It



キャンペーン 新品未使用 Celsus Sound Sp One P パッシブタイプ 2本1組 スピーカー セルサスサウンド Spone P Celsus Spone P モバックス 通販 Yahoo ショッピング




Sp One P Celsus Sound Hifi Do Mcintosh Jbl Audio Technica Jeff Rowland Accuphase




Hybridization Mixing Of Two Or More Atomic Orbitals




Celsus Sound Sp One が新色を加えて販売再開予定です



Celsus Sound Sp One P 월간 오디오




Celsus Sound Sp One Active And Passive Audio Graffiti



Plos One P Coumaric Acid Influenced Cucumber Rhizosphere Soil Microbial Communities And The Growth Of Fusarium Oxysporum F Sp Cucumerinum Owen




新品未使用 Celsus Sound Sp One P パッシブタイプ 2本1組 スピーカー セルサスサウンド Spone P 携帯 スマホの激安販売通販ショップ モバックス Mobax 新品 中古




New Sp For Acer Aspire One 522 533 532g Ao532h 532h 521 Ao522 D260 D270 Spanish Laptop Keyboard Replacement Keyboards Aliexpress




Abarth Una Splendida One Off Omaggia La 1000 Sp Degli Anni Sessanta La Stampa




One Loop Contribution Of The S And P Left Panel And The V And A Download Scientific Diagram




画像をダウンロード Sp Onep 無料のワンピース画像




Simple Plan Facebook




Celsus Sp One Active Speakers Review Headfonics Com




Aerie Swimsuit One Piece S P One Piece Swimsuit Swimsuits One Piece




Raccolta Ar Da 381 A 390 Audiovideoteam




Hivi夏のベストバイ19 スピーカー部門 1 ペア10万円未満 Stereo Sound Online




Lecture 16 C 1403 October 31 05 18




Astell Kern Acro L1000 Celsus Sound Sp One P Kupit V Kieve I Ukraine Po Cene Grn Na Portativ Ua




Bel Art Sp Scienceware Single Piece Buchner Funnels 1000ml Capacity 122mm Plate Diameter 104mm Rim To Plate Bel Art Sp Scienceware Single Piece Buchner Funnels Fisher Scientific




Applicazione Mobile Di Sap Business One



Chapter 9 Notes Ap Chemistry Galster Molecular Geometry




Arena Sp Der One P Ece Girls Swimwear Superdokan




Battle Captain Smoker Vs Captain Kuro One Piece Fighting Path The Bloodline Android Gameplay Youtube




Onerepublic Padua 03 05 22 21 30 Tickets Ticketone




Abarth Una Splendida One Off Omaggia La 1000 Sp Degli Anni Sessanta La Stampa



画像をダウンロード Sp Onep 無料のワンピース画像




Sp One Piper Pa 34 2t Seneca Iii Private Bogdan Jankowiak Jetphotos




Termokamera Flir One Pro Android Usb C 435 0007 03 Sp 160 X 1 Pix Ed System A S




Pardini Sp Hp All In One Pardini Armi



Chapter 10 Chemical Bonding Molecular Shapes Valence Bond



1 Chemical Bonding 2 Lewis Theory An Overview Valence E Play A Fundamental Role In Chemical Bonding E Transfer Leads To Ionic Bonds Sharing Of Ppt Download




Two Oocysts Of Eimeria Sp One Viable And Sporulating On The Left Download Scientific Diagram
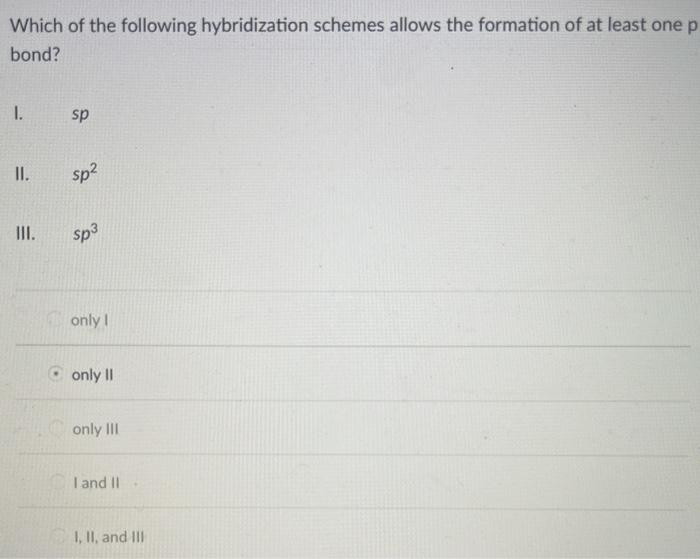



Solved Which Of The Following Hybridization Schemes Allows Chegg Com




Sp One P Celsus Sound Hifi Do Mcintosh Jbl Audio Technica Jeff Rowland Accuphase




A Sp Hybridisation One S Orbital Mixes With One P Orbital Forming Two Equivalent Sp Hybrid Orbitals Types Of Hybridisation Linear 180 Two Sp Hybrid




Fabio Manucci Cenozoic Scenes




Automatic One Layer Cutter Cutting Width 170 Cm Texi Xyz Sp 170



画像をダウンロード Sp Onep 無料のワンピース画像



Jstor Org




Sp One Piper Pa 34 2t Seneca Iii Private Lukasz Stawiarz Jetphotos




Solved Which One Of The Following Statements Is False A An Chegg Com




Acrol1000 Instagram Posts Photos And Videos Picuki Com




Abarth Una Splendida One Off Omaggia La 1000 Sp Degli Anni Sessanta La Stampa



0 件のコメント:
コメントを投稿